Statistics in Medicine — Reporting of Subgroup Analyses in
Descrição
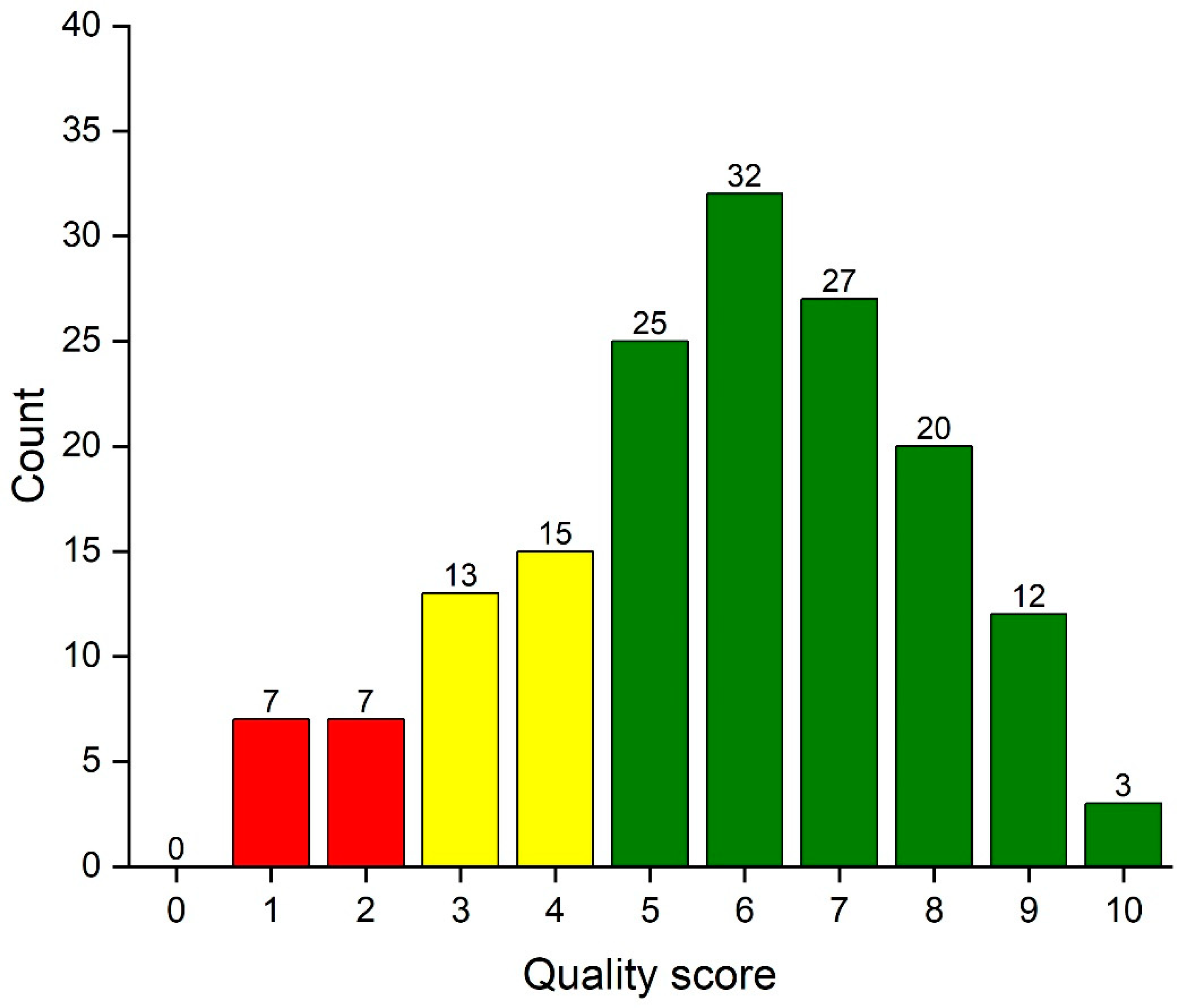
Applied Sciences, Free Full-Text
If you spend enough time following biotech, you’ll encounter a common situation: A big clinical trial fails, but the company points to a glimmer of an

Subgroup analysis: How to evaluate post hoc tests for significance in failed clinical trials

A critical review of graphics for subgroup analyses in clinical trials - Ballarini - 2020 - Pharmaceutical Statistics - Wiley Online Library

Subgroup analyses for matched cohorts (unadjusted). P values for

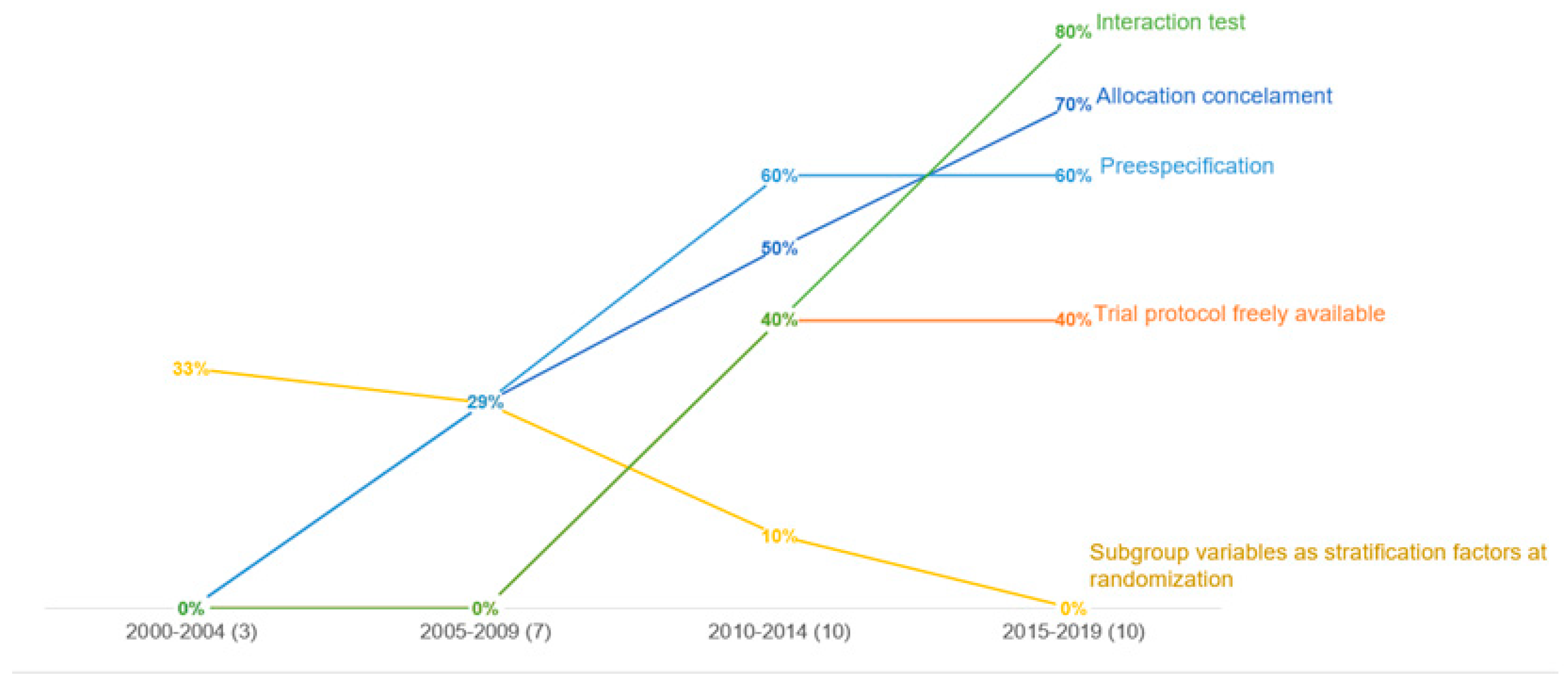
Statistics in Medicine — Reporting of Subgroup Analyses in Clinical Trials
JPM, Free Full-Text

Full article: Tezepelumab compared with other biologics for the treatment of severe asthma: a systematic review and indirect treatment comparison

Subgroup analyses in randomized phase III trials of systemic treatments in patients with advanced solid tumours: a systematic review of trials published between 2017 and 2020 - ESMO Open

Disease progression and treatment response in data-driven subgroups of type 2 diabetes compared with models based on simple clinical features: an analysis using clinical trial data - The Lancet Diabetes & Endocrinology

Statistical Considerations for Subgroup Analyses - ScienceDirect
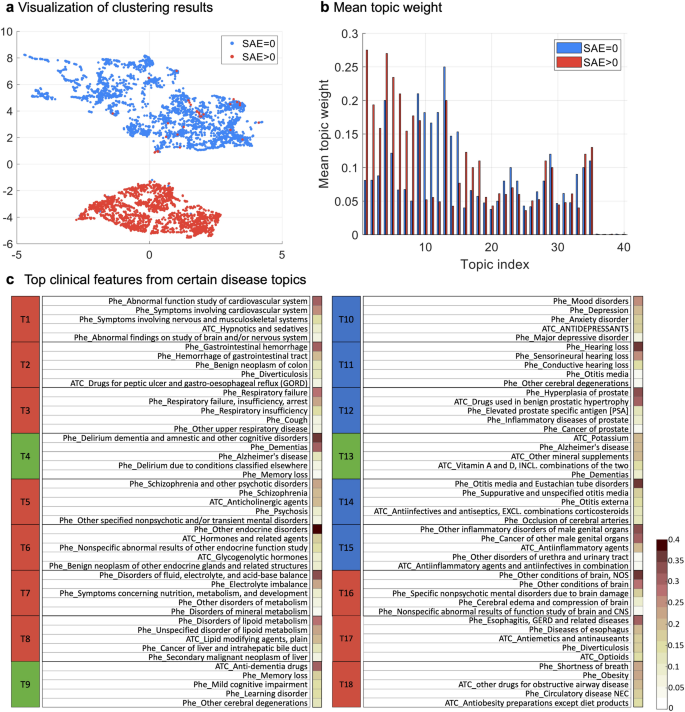
Machine learning enabled subgroup analysis with real-world data to inform clinical trial eligibility criteria design
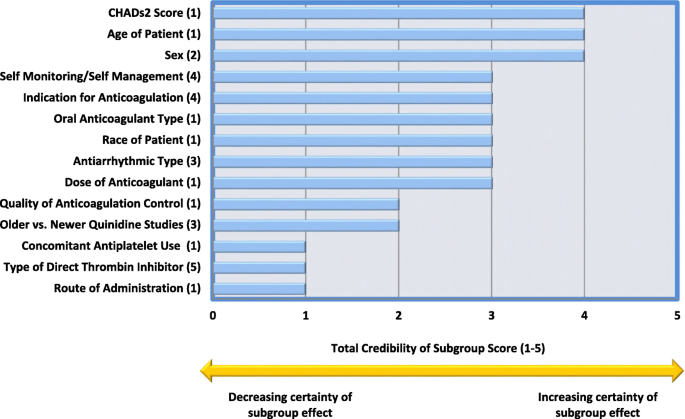
A meta-epidemiological study of subgroup analyses in cochrane systematic reviews of atrial fibrillation, Systematic Reviews
de
por adulto (o preço varia de acordo com o tamanho do grupo)